ViSi Mobile System General Information
What is the ViSi Mobile System?
The ViSi Mobile Surveillance Monitoring System is a patient-worn, wireless vital sign monitoring platform that is designed to detect patient deterioration (including life-threatening arrhythmias, vital sign changes, and patient falls) and alert bedside staff earlier without causing alarm fatigue. The system is connected to your hospital’s wireless infrastructure to allow:
a. Your patient’s information is to be sent to the wrist device for easier patient assigning
b. Data for all patients on the monitoring system is to be displayed at a central location
c. Patient data is to be sent directly to the patient’s medical record
d. Alarms indicating patient deterioration are to be sent directly to the staff phones
What is the Indication for Use?
The ViSi Mobile Monitoring System is intended for use by clinicians and medically qualified personnel for single or multi-parameter vital signs monitoring of adult patients. The ViSi Mobile System has a CE mark and is FDA cleared for Non-Invasive Blood Pressure (NIBP) monitoring (single cuff inflation) and continuous monitoring of Heart/Pulse Rate, SpO2, Respiration Rate, and Skin Temperature. The system also has patented technology that allows for Continuous Non Invasive Blood Pressure (cNIBP), which has not previously been available for general care floors. Additionally, it can display a continuous ECG waveform and can detect and alarm for atrial fibrillation, ventricular tachycardia, ventricular fibrillation, and asystole.
What is the intended patient population?
Adults age 18 or over
What vital signs are captured by the ViSi Mobile System?
Vital Signs captured by ViSi Mobile System, include the following:
- SpO2 / Pulse Rate
- ECG / Heart Rate
- Respiration Rate
- Skin Temperature
- Non-invasive Blood Pressure (NIBP)
- Continuous Non-Invasive Blood Pressure (cNIBP)
What disposables are required to operate the ViSi Mobile System?
There are 5 single, patient-use components:
• Wrist cradle and strap
• Chest Cable securement set
• Disposable ECG Electrodes (recommend 3M™)
• Blood Pressure Cuff (validated with Welch Allyn® Flexiport™ & GE Critikon™ Soft-CufT™)
• Thumb wrap/Thumb tape
How is the ViSi Mobile System powered?
Both the Wrist Transceiver and Cuff Module are powered by internal rechargeable Li-Ion batteries that are recharged in either an 8-bay or 2-bay charger depending on the bedside workflow. The Wrist Transceivers last 12-14 hours, and the Cuff Module will need to be recharged after approximately 30 inflations. Both will take approximately 4 hours to charge from fully depleted.
What does the setup look like on a patient?
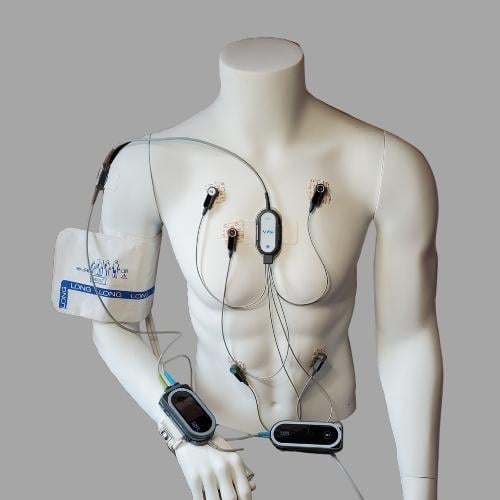
Cuff is only applied during initial application and calibration.
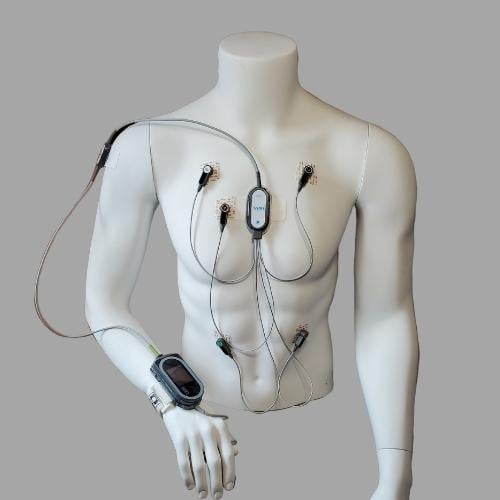
Once the system has been set up and calibrated, the cuff is removed.
Does the patient need to wear a cuff during cNIBP?
There are several situations when the patient must wear the blood pressure cuff. The cuff is utilized to establish the initial patient-specific calibration. Following a successful calibration, the cuff can be removed, and the ViSi Mobile System will display beat-to-beat blood pressure continuously without the need for cuff inflations. If the system needs to be recalibrated, the cuff will be needed.
How does the ViSi Mobile System cNIBP work?
ViSi’s patented technology allows for continuous blood pressure to be measured non-invasively on a beat-to-beat basis without the need for frequent cuff inflations. The system first detects the onset of the R wave via the chest sensor and then measures the time it takes for the resulting pulse to be detected by the thumb sensor. Once it has determined the average time for the pulse to arrive at the thumb sensor (AKA “pulse arrival time” or PAT), the system prompts the user to obtain an initial “calibrating cuff measurement” and uses that measurement along with the PAT to calculate pulse wave velocity and convert the measurements into a usable blood pressure in mmHg(or kPa). Once the calibrating cuff measurement is obtained, the cuff can be removed, the patient’s beat-to-beat blood pressure is displayed at the wrist, and the RVD and the value is refreshed every 3 seconds for real-time measurements.
What is the accuracy of the ViSi Mobile System cNIBP?
The ViSi Mobile System cNIBP technique has been compared in clinical trials to invasive arterial lines and has shown accuracy in these studies within five mmHg. The systolic pressure increases as a pulse wave travels down the arterial system. This is noticeable when comparing systolic blood pressure measured via an invasive arterial line versus auscultation. To adjust for this change in systolic pressure, the blood pressure obtained during cuff calibration for the cNIBP is set to calibrate against the gold standard of a manual blood pressure (auscultatory method).
How often does cNIBP require calibration?
• If the system detects a Significant change in Mean Arterial Pressure (alert)
• If the system detects that a sensor has been disconnected for an extended period of time (alert)
• If a vasopressor has been administered, we recommend recalibration (no alert)
• Required every 24 hours to ensure accuracy (alert)
Do I lose cNIBP for patients that I calibrated more than 24 hours ago?
A “Calibrate cNIBP” alert will display on the monitor & RVD if the last known cNIBP calibration was more than 24 hours ago. cNIBP will continue to display with the alert until the next device swap. If a clinician sees the alert during device swap, cNIBP must be recalibrated. If the alert is not present at the time of the device swap, the calibration will transfer to the next monitor.
Can the patient access the monitoring functions?
We recommend enabling an optional clinical authentication code to prevent patient tampering and avoid the access of PHI. The patient will only be able to view their own vital signs, waveforms, and menu options (only accessible using the clinical authentication code).
Can the patient shower while wearing the ViSi Mobile System?
All components, with the exception of the Cuff Module, are waterproof and can be submerged in water. Because the water on the patient’s skin will cause noise in the vital sign measurements and potentially false alarms, it is not recommended that the patient shower while wearing the ViSi Mobile System. Follow the Pause Monitoring workflow outlined in the User Manual if the patient will need to shower or bathe.
Will the monitor identify pacemaker spikes?
Yes, the Monitor’s ECG channel is capable of monitoring patients with an Implanted Pacemaker (PM), Implantable Cardioverter-Defibrillator (ICD), or Cardiac Resynchronization Therapy (CRT) device. A vertical dashed line before the P wave (atrial pace), and before the QRS (ventricular pace) indicate paced events. In the case of biventricular pacing (CRT), two vertical dashed lines occur before the QRS.
Can the Monitor be used during defibrillation?
Yes, the system can be used during defibrillation without damaging or affecting the system. A Heart Rate reading will be displayed within 20 seconds after a defibrillation event. An ECG waveform will be displayed within 5 seconds after a defibrillation event.
Does the ViSi Mobile System have bedside alarm capabilities?
The patient-worn ViSi Mobile monitor itself is the primary alarming device. When integrated with a hospital’s wireless network, the alarms are then propagated to the Remote Viewer Display (central monitor) and will only alarm at the wrist monitor if the Wi-Fi connection is unavailable.
What is the cleaning and disinfecting procedure for the ViSi Mobile System?
To clean the ViSi Mobile Monitoring System components
• Hand wash the System components using mild soap or detergent (e.g. Alconox) and water
• Cuff module is NOT waterproof, so clean it with a damp cloth
• Use moistened cloths to remove residual cleaner
• Dry the System components
• Visually examine each System component to ensure all soil contaminants have been removed
To disinfect the ViSi Mobile System Components
• follow the recommended application of the disinfectant or use a basic wipe moistened with 70% isopropyl alcohol
• Avoid hydrogen peroxide and/or bleach (see user guide for additional details)
• Dry thoroughly
Does the Monitor have clinical and technical alarm capabilities?
Yes. The ViSi Mobile System has been designed to reduce the number of non-actionable alarms per patient. Clinical alarms will annunciate based on configured parameter thresholds and delays set by the clinical staff. The majority of technical alarms (alerts) will only visually annunciate.
Are Sotera and Sotera Wireless the same company?
Yes. In June of 2022, Sotera Wireless, Inc. filed for a fictitious business name to be able to use Sotera, Inc. (without Wireless) to conduct business. Both Sotera and Sotera Wireless are legally acceptable names for our organization. You may also see the company referred to as Sotera Digital Health, Digital Health is a tagline.
ViSi Mobile System Architecture / Infrastructure and Network Connectivity
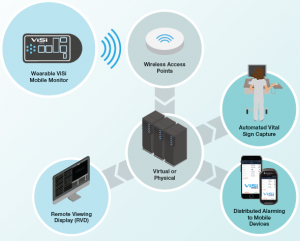
Do you have a network topology diagram?
How does the ViSi Mobile Monitor integrate with a hospital network?
The ViSi Mobile Monitors integrate with a hospital’s existing 802.11 networks. While there are additional interoperability options available for distributive alarming, the standard system consists of 3 main components:
• ViSi Mobile Monitor – Worn by the patient and communicates with the ViSi Mobile Appliance over the wireless network using an 802.11 radio.
• ViSi Mobile System Software – Installed on a virtual server; the System software compiles and distributes patient information to the ViSi Mobile Remote Viewer(s).
• ViSi Mobile Remote Viewing Display (RVD) – Central station that allows the user to view patient vital signs and other continuous data
What co-existence issues with other products might arise if a hospital’s wireless network provides connectivity to multiple devices of differing manufacturers?
The ViSi Mobile Monitor can coexist with various medical and enterprise devices based on our lab and live testing. Our co-existence testing involved over 20 different yet common medical devices from differing manufactures.
ViSi Mobile Device and Remote Viewing Display
Is the application HIPAA compliant?
Yes. The application isolates PHI from patient vital sign information for HIPAA compliance.
Can the ViSi Mobile Server be deployed virtually?
Yes, it can be deployed on a virtual machine. An onsite assessment and validation will be required.
Does the ViSi Mobile Server require remote secure VPN access?
Yes, remote access via SecureLink is required for the ViSi Mobile Application (Server) for troubleshooting and software updates.
How will Sotera manage Windows updates?
Sotera Customer Support will coordinate with the site’s technical resources to implement software updates of the RVD workstation, software configuration, or updates to the Windows Operating System (OS). However, the customer is responsible for validating in a non-production environment. This technical assistance is included with a Sotera System Support Agreement.
Are there known conflicts with Anti-Virus products and /or scans?
Although there are no known conflicts, the customer should validate the system functionality in combination with the preferred Anti-Virus product.
ViSi Mobile System Interoperability
How long will interoperability projects take from start to go live?
It varies. Sotera offers numerous integration options to align with your interoperability strategy. Our discovery process will be the start of the information exchange that will help to identify the appropriate solution for your IT environment and clinical workflows. As an output of the discovery process, a Statement of Work will be developed.
What equipment is needed for non-production testing for integrations?
The ViSi Mobile monitor and sensors are required along with disposables. A charger is recommended to ensure the device is charged. A virtual server and RVD are set up by Sotera.
What is the interoperability process?
Although each integration project will be different, the phases below identify the high-level interoperability milestones within the Sotera Digital Health Implementation process. The Sotera Interoperability Engineer will assist with implementing your organization’s integration options.
Phase 1: Interoperability Discovery
• Align project requirements and objectives
• Perform Gap Analysis
• Set Project timelines
Phase 2: Establish Connectivity
• Establish SecureLink communication
• Install a virtual non-production environment
• Perform required Sotera software upgrades
• Record Sotera clinical configuration and apply it to the system
Phase 3: ADT (HL7 Patient Demographics)
• Establish ADT connectivity
• Build ADT
• Functional Testing of ADT
• Integrated Interface Testing (UAT) by Clinical Users (if required)
Phase 4: EMR (HL7 Vital Signs Results aka ORU)
• Establish EMR connectivity
• Build EMR Interface
• Functional Testing of EMR Interface
• Integrated Interface Testing (UAT) by Clinical Users (if required)
Phase 5: Distributed Alarming (HL7)
• Establish Distributive Alarming Connectivity
• Build Interface
• Functional Testing of Distributive Alarming
• Integrated Testing (UAT) of Distributive Alarming (if required)
Phase 6: Production Activation
• Sotera will work with your application team to confirm the change window for production migration.
• During the prescribed change window, Sotera will migrate the configuration to production.
• Sotera will request Customer Interoperability Acceptance signoff once testing has been completed.
ADT Configurations
Can the ViSi Mobile System interface with an ADT server?
Yes, the ViSi System can interface with ADT. The inbound ADT data is utilized by the Sotera application to provide patient identifiable information for display on the ViSi Mobile Monitors.
EMR and Distributive Alarming Integrations
With which EMRs can ViSi Mobile System integrate?
The ViSi Mobile System can integrate with most EMR systems through HL7 or via a middleware like Capsule. Active integrations include Epic, Cerner, Meditech and HMS/Medhost.
With which Distributive Alarming Systems can the ViSi Mobile System integrate?
The ViSi Mobile System can integrate with most distributive alarming middleware. Active integrations include Extension, Cerner and Connexall.
Do the interfaces support transfer of waveforms?
The ViSi Mobile System is capable of sending raw waveform data to a receiving system.
What iBus version is require for Cerner integration?
For PowerChartTM, Cerner requires iBus 5.1 or higher. For ConnectTM and AlertLinkTM, Cerner requires iBus 5.2.6 or higher. ViSi Mobile System is compatible with these versions of the iBus.
How is the association made between the ViSi Mobile Monitor and the Cerner iBus?
Sotera Wireless has an interface through iBus utilizing Cerner’s advanced bidirectional Patient to Device Association (P2DA). The advanced P2DA workflow reduces numerous inefficiencies with device connectivity in that the monitor only needs to be associated a single time and not each time a battery swap is complete or after the session is paused and restarted.